 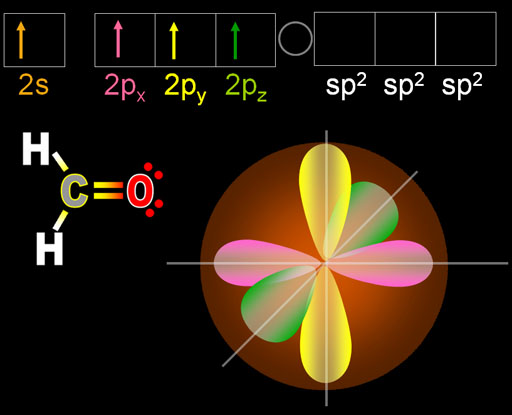
Their names indicate the orientation of the orbitals in space and the designation (sp 2, sp 3, etc) indicates the number and types of atomic orbitals involved in hybridization. Tetrahedral or sp 3 hybridization e.g.The hybridization involving s and p orbitals are of the following three types: Types of Hybridization and Geometry of Molecules: The hybrid orbitals have maximum symmetry and definite orientation in space so that the mutual force of repulsion of electrons is avoided.The hybrid orbitals are concentrated in one particular direction to achieve greater overlapping.Similar to atomic orbitals, each hybrid orbital can have a maximum of two electrons.The shape of the hybrid orbitals is different from that of the original atomic orbital.The hybrid orbitals may differ from one other in their orientations.All hybrid orbitals are identical with respect to energy and directional character.The total number of hybrid orbitals formed is equal to the number of atomic orbitals involved in the hybridization process.For a tetrahedrally coordinated carbon (e.g. Thus 2s and 2p can hybridize, 3s and 3p can also hybridize, but 2s and 3p cannot. Hybridisation describes the bonding of atoms from an atoms point of view. Atomic orbitals participating in hybridization should have nearly the same energy.It can also be noted that it is not possible for the 3f orbital to exist because that would require. The names of these atomic orbitals will be 3s (for n3 and l0), 3p (for n3 and l1), and 3d (for n3 and l2). Atomic orbitals undergoing hybridization should belong to the same atom or ion. For example, if the value of ‘n’ is equal to 3, the possible values of ‘l’, which range from zero to (3-1), are 0, 1, and 2.Note: The above paragraphs give limitations of the valence bond theory.Ĭharacteristics or Rules of hybridization: It can be explained on the basis of hybridization. all the four C-H bonds in methane molecule are equivalent in terms of strength, energy, etc. To explain The equivalence of bonds we have to use the concept of a process of mixing and recasting of atomic orbitals. 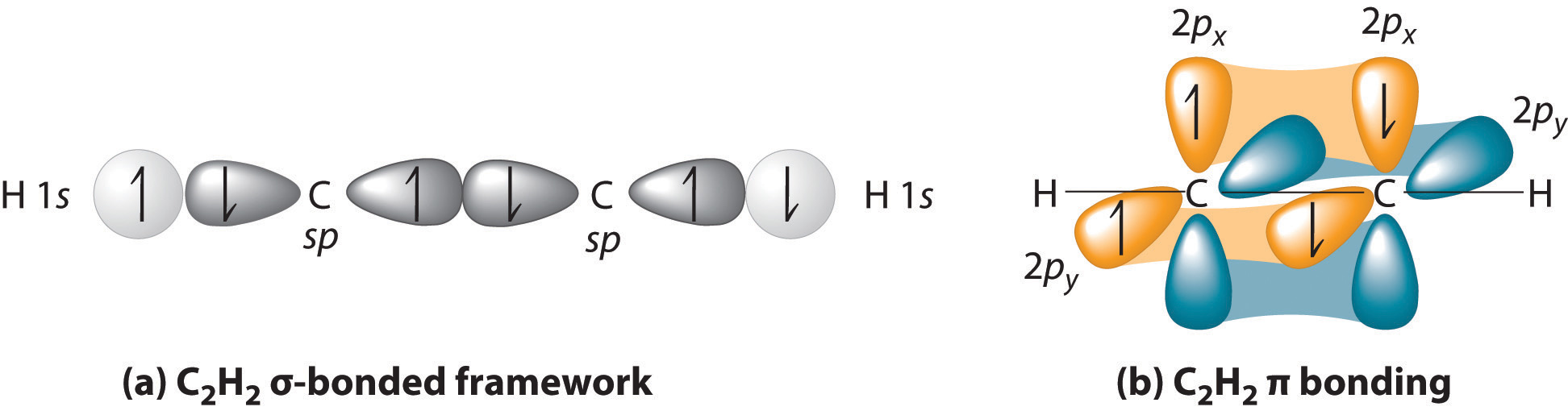
Valence bond theory failed to explain this change. But the measured bond angle is 104.3° and the molecule is V-Shaped. in the formation of H 2O molecule, the H – O – H bond angle should be 90°. 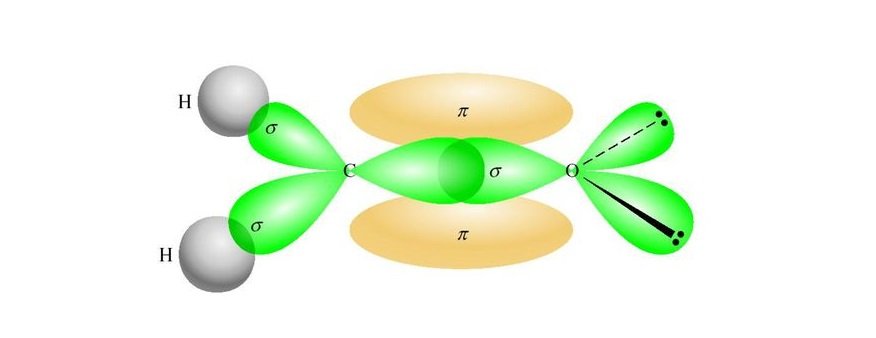
The s orbital and all three p orbitals have been mixed, thus the hybridization is sp 3. Valence Bond Theory fails to explain the observed geometry of the molecules of water and ammonia e.g. In order to form four hybrid orbitals, four atomic orbitals have been mixed. The four valence atomic orbitals from an isolated carbon atom all hybridize when the carbon bonds in a molecule like CH4 with four regions of electron density. Valenceīond theory fails to explain this phenomenon. However, theseĮlements form the compounds having valency 2, 3, and 4 respectively. Zero-valent, monovalent and divalent respectively. Thus Beryllium, Boron, and Carbon should be The sp hybrid orbitals of carbon occur in compounds such as ethyne that contain a triple bond. The valence orbitals of an atom surrounded by a tetrahedral arrangement of bonding pairs and lone pairs consist of a set of four sp 3 hybrid orbital.Valence bond theory valency of an element depends on a number of unpairedĮlectrons in the orbitals. A lone pair, an unpaired electron, a single bond, or a multiple bond would each count as one region of electron density. As we know from the discussion of VSEPR theory, a region of electron density contains all of the electrons that point in one direction. The central atom(s) in each of the structures shown contain three regions of electron density and are sp2 hybridized. Valence bond theory would predict that the two \ce.įigure 7.5.9. Oxygen has the electron configuration 1 s 22 s 22 p 4, with two unpaired electrons (one in each of the two 2p orbitals). As an example, let us consider the water molecule, in which we have one oxygen atom bonding to two hydrogen atoms. 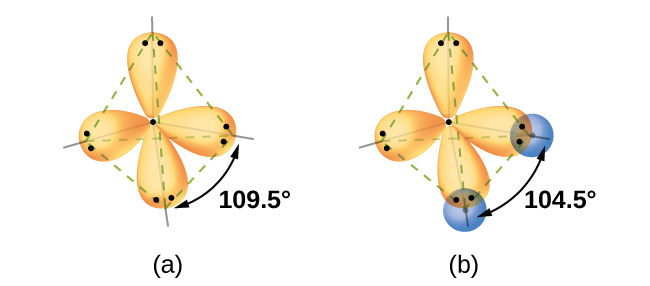
However, to understand how molecules with more than two atoms form stable bonds, we require a more detailed model. Thinking in terms of overlapping atomic orbitals is one way for us to explain how chemical bonds form in diatomic molecules. This is not consistent with experimental evidence. The hypothetical overlap of two of the 2p orbitals on an oxygen atom (red) with the 1s orbitals of two hydrogen atoms (blue) would produce a bond angle of 90°. Determine the hybrid orbitals associated with various molecular geometriesįigure 7.5.1.Explain the concept of atomic orbital hybridization. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
